
Greg Springsteen
Professor, Chemistry
- Email: [email protected]
- Phone: 864.294.2763
- Office: Plyler 236E, Townes Science Center
Greg Springsteen is a chemistry educator, researcher, and innovator who has collaborated with over 50 undergraduate researchers in his lab in the fields of prebiotic chemistry and metabolism. He received a Henry Dreyfus Teacher-Scholar Award in 2018, as one of eight faculty nationwide who demonstrated original scholarly research of outstanding quality, as well as excellence and dedication in undergraduate education. Greg has also served for over a decade as a member and Theme Leader in the Center for Chemical Evolution, a 100-scientist NSF- and NASA-funded research consortium. Research from his group has been recognized nationally in journals including Angewandte Chemie, Nature Communications, and Nature Chemistry.
The Springsteen group is also heavily involved in entrepreneurship and has submitted two provisional patents, one US non-provisional, one PCT application. Work in the group has also been commercialized by the founding of a company (Aconabolics LLC) focused on the development of labeled-keto and organic acids for use in metabolic research and diagnostics. Greg also serves as a faculty representative on Furman’s Innovation and Entrepreneurship Leadership council.
Since arriving at Furman in 2007, Greg has taught seven Lecture Courses including Organic Chemistry, BioOrganic Chemistry/Org. II, General Chemistry, Biochemistry, Advanced Biochemistry, Molecular Gastronomy, and Techniques in Chemistry, along with their laboratory components, and was awarded the 2018 South Carolina Independent Colleges and Universities teaching award, which honors the “Best of the Best among the faculty at the twenty member colleges and universities” based on efforts “that demonstrate its institution's highest standards in teaching.”
Education
-
Postdoctoral Training, The Scripps Research Institute
- Ph.D., North Carolina State University
- B.A., University of Virginia
Research
In my research lab, a group of 3-6 undergraduates and I seek to understand the chemical origins of life (abiogenesis). We search for answers by comparing the mechanistic organic chemistry of biological metabolism with that of spontaneous geochemical and astrochemical reactions. Our efforts are guided by the hypothesis that core components of modern metabolism existed as spontaneous reactions in an abiotic (without life) environment, prior to their incorporation and adaptation into chemical systems capable of rudimentary replication. These central biosynthetic “arteries” may then have remained frozen at the core of an expanding metabolic network with increasing functionality. If true, the discovery of abiotic environments that reproduce these arteries will aid in the recapitulation of abiogenesis, much as an analysis of the US interstate highway system might reveal important clues about the pathways of the early emigrant trails that served as their foundation.
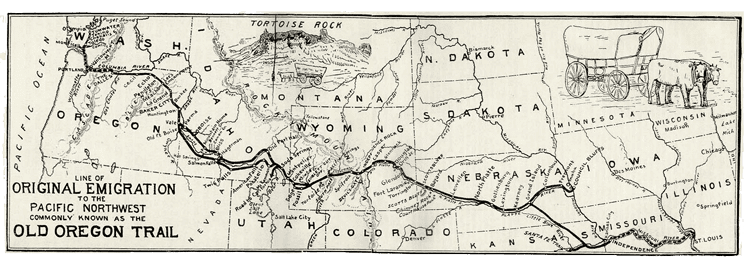 Courtesy of the University of Texas Libraries. The University of Texas at Austin.
Courtesy of the University of Texas Libraries. The University of Texas at Austin.
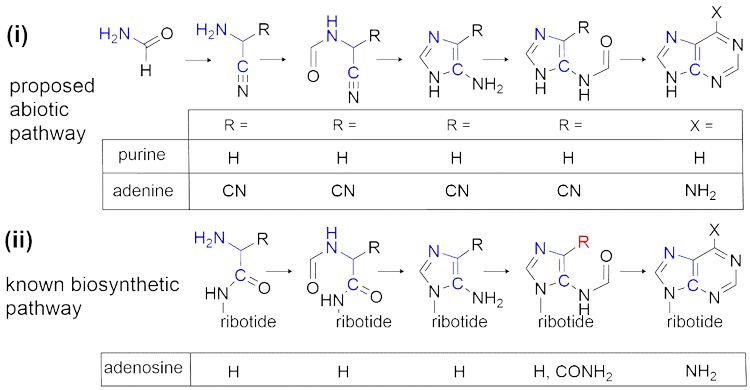
We are particularly interested in the mechanisms of formation of nucleic acid components (DNA/RNA), and oxidative decarboxylation pathways like the citric acid cycle (TCA). Our primary methods of discovery include the synthesis of isotopically labelled intermediates for tracing protometabolic reaction pathways, and the recovery and analysis of intermediates from plausible prebiotic reaction mixtures. We use data obtained from these efforts to reconstruct step-by-step abiotic synthetic mechanisms and compare these routes to modern metabolism.
Please view our publications (above tab) and those of our CCE collaborators) to explore recent discoveries towards our understanding of the chemical origins of life.
-
Pulletikurti, S.; Yadav, M.; Springsteen, G.; Krishnamurthy, R. "Prebiotic synthesis of α-amino acids and orotate from α-ketoacids potentiates transition to extant metabolic pathways", Nature Chemistry, 2022, 14, 10, 1142-1150.
-
Clay, A.P.; Cooke, R.E.; Kumar, R.; Yadav, M.; Krishnamurthy, R.; Springsteen, G. "A Plausible Prebiotic One‐Pot Synthesis of Orotate and Pyruvate Suggestive of Common Protometabolic Pathways", Angewandte Chem Int. Ed., 2022, 61, 11.
-
Stubbs, R. T; Yadav M., Krishnamurthy, R.; Springsteen, G. "A plausible metal-free ancestral analogue of the Krebs cycle composed entirely of α-ketoacids", Nat. Chem., 2020, 12, 1066-1022.
-
Springsteen, G.; Yerabolu, J. R.; Nelson, J.; Rhea, C. J.; Krishnamurthy, R. Linked cycles of oxidative decarboxylation of glyoxylate as protometabolic analogs of the citric acid cycle. Nature Communications, 2018, 9, 91.
-
Rice, G. B.; Yerabolu, J. R.; Krishnamurthy, R.; Springsteen, G. The Abiotic Oxidation of Organic Acids to Malonate. Synlett 2017, 28 (1), 98-102.
-
Springsteen, G. Reaching Back to Jump Forward: Recent Efforts towards a Systems-Level Hypothesis for an Early RNA World. ChemBioChem 2015, 16 (10), 1411-1413.
-
Wang, J.; Gu, J.; Nguyen, M. T.; Springsteen, G.; Leszczynski, J. From formamide to purine: a self-catalyzed reaction pathway provides a feasible mechanism for the entire process. J. Phys. Chem. B 2013, 117, 9333-42.
-
Wang, J.; Gu, J.; Nguyen, M. T.; Springsteen, G.; Leszczynski, J. From formamide to adenine: a self-catalytic mechanism for an abiotic approach. J. Phys. Chem. B 2013, 117, 14039-45.
-
Wang, J.; Gu, J.; Nguyen, M. T.; Springsteen, G.; Leszczynski, J. From formamide to purine: an energetically viable mechanistic reaction pathway. J. Phys. Chem. B 2013, 117, 2314-20.
-
Hudson, J. S.; Eberle, J. F.; Vachhani, R. H.; Rogers, L. C.; Wade, J. H.; Krishnamurthy, R.; Springsteen, G. A unified mechanism for abiotic adenine and purine synthesis in formamide. Angew. Chem., Int. Ed. Engl. 2012, 51, 5134-7.